How Does Laser Cleaning Work in 5 Steps
Laser cleaning is an eco-friendly process used to remove rust, paint, oxide and other contaminants from metal surfaces. Because of its efficiency, it is being used in an increasing number of applications.

Surface preparation is essential to ensure the success of adhesive bonding. It involves optimizing the surface of the substrate for a stronger bond with the adhesive.
There are three primary aspects to surface preparation: removing contaminants (dust, grease, oil, rust, etc.), roughening the surface, and modifying the chemical composition.
In this article, you’ll learn how to prepare surfaces to create a strong bond for adhesives. You will also learn how lasers, abrasives, chemicals, and plasma affect surfaces. Finally, you will get an overview of the tests performed to validate the quality of surface preparation.
But first, let’s go over the concept of surface free energy, which is at the heart of how materials bond together.
To maximize the bonding quality between adhesives and other materials, it’s important to understand how materials bond together.
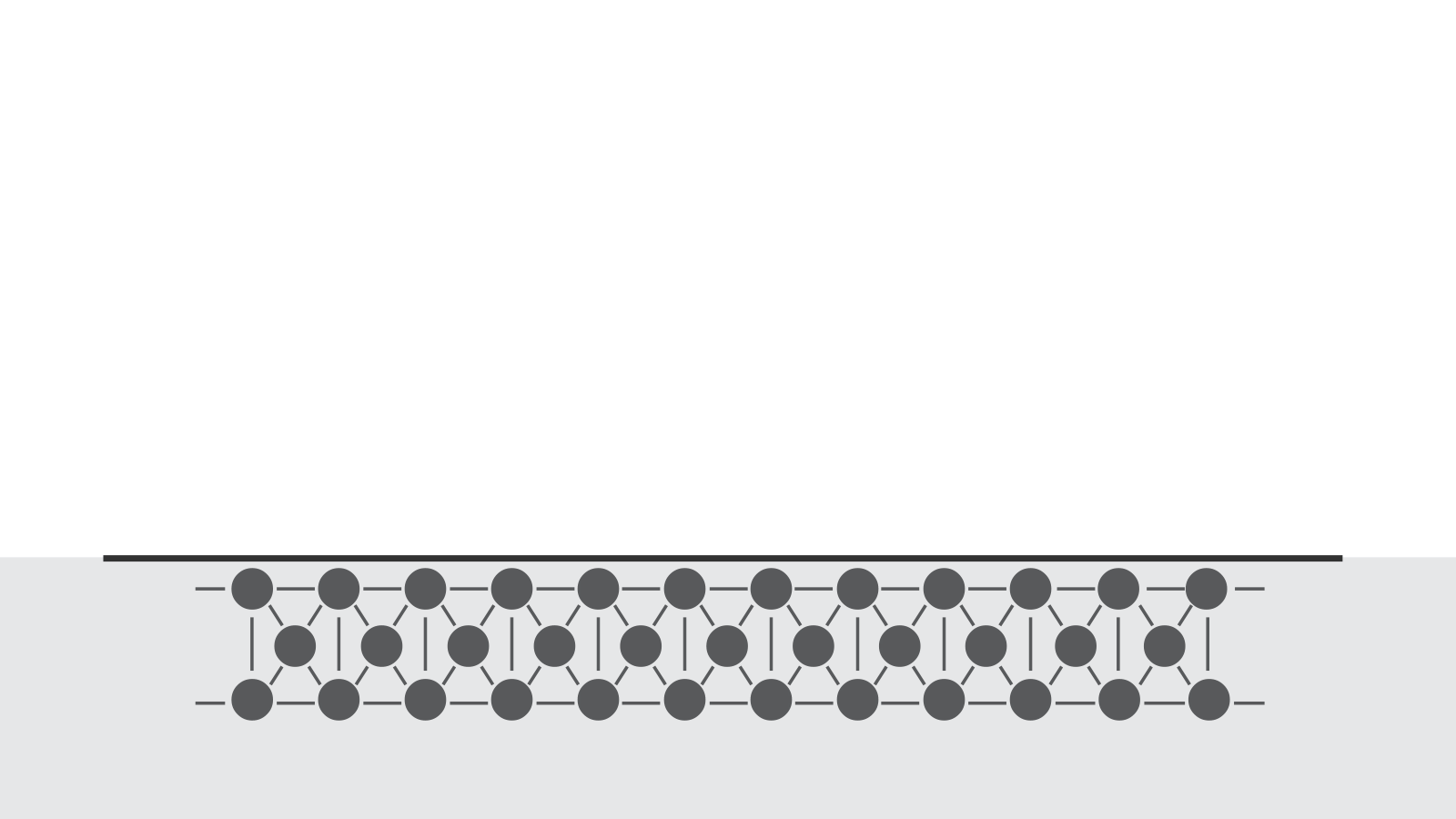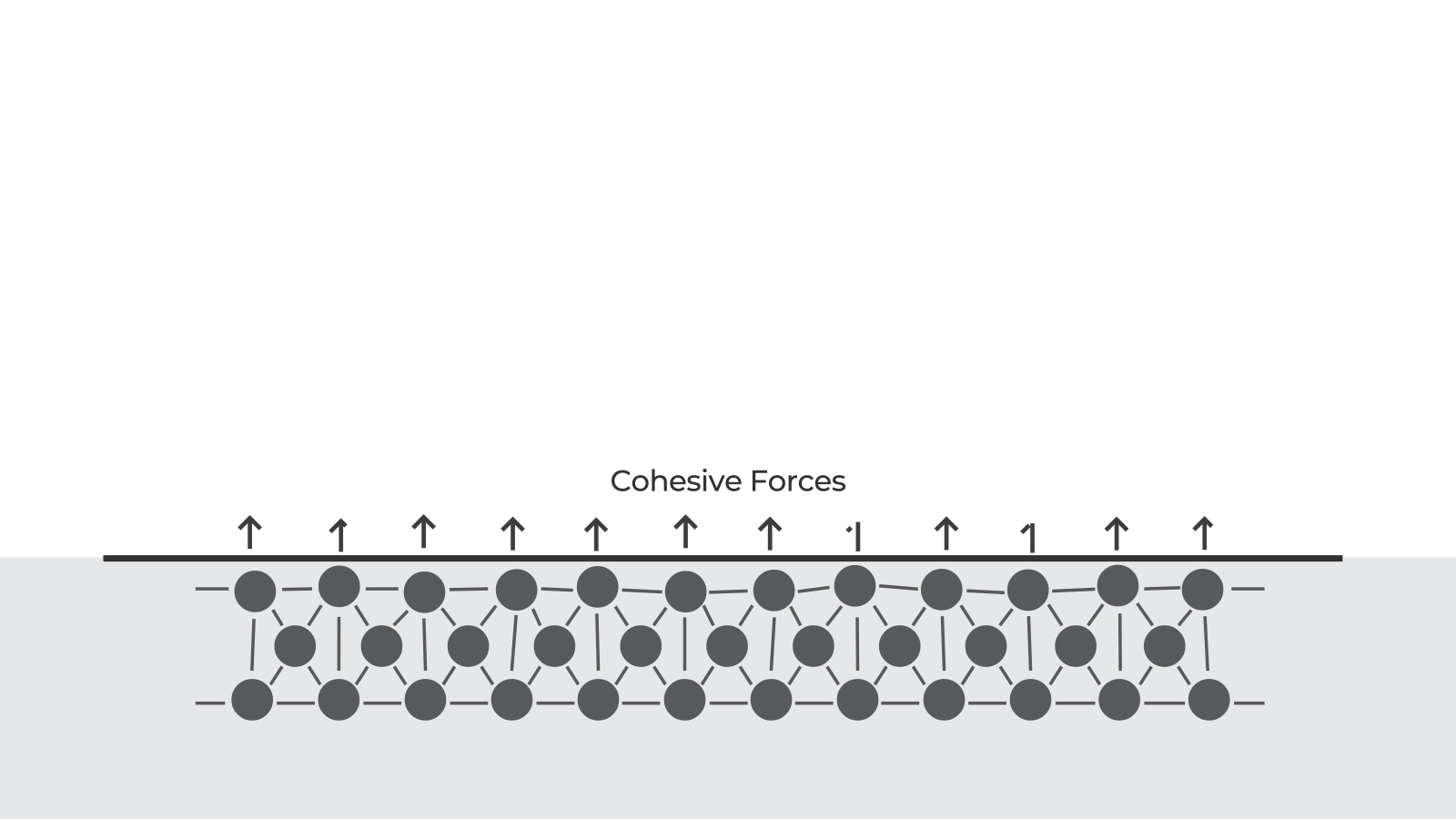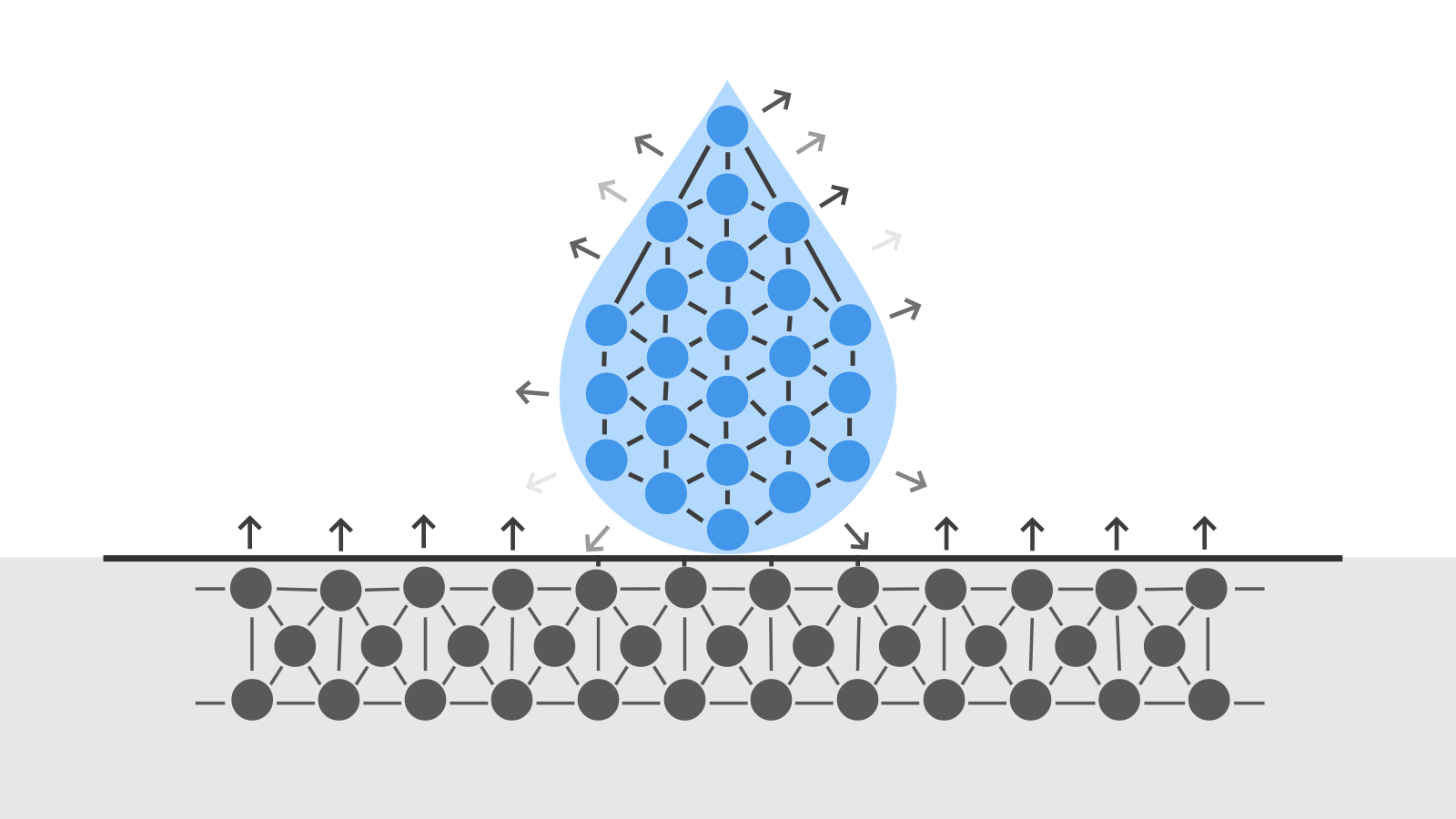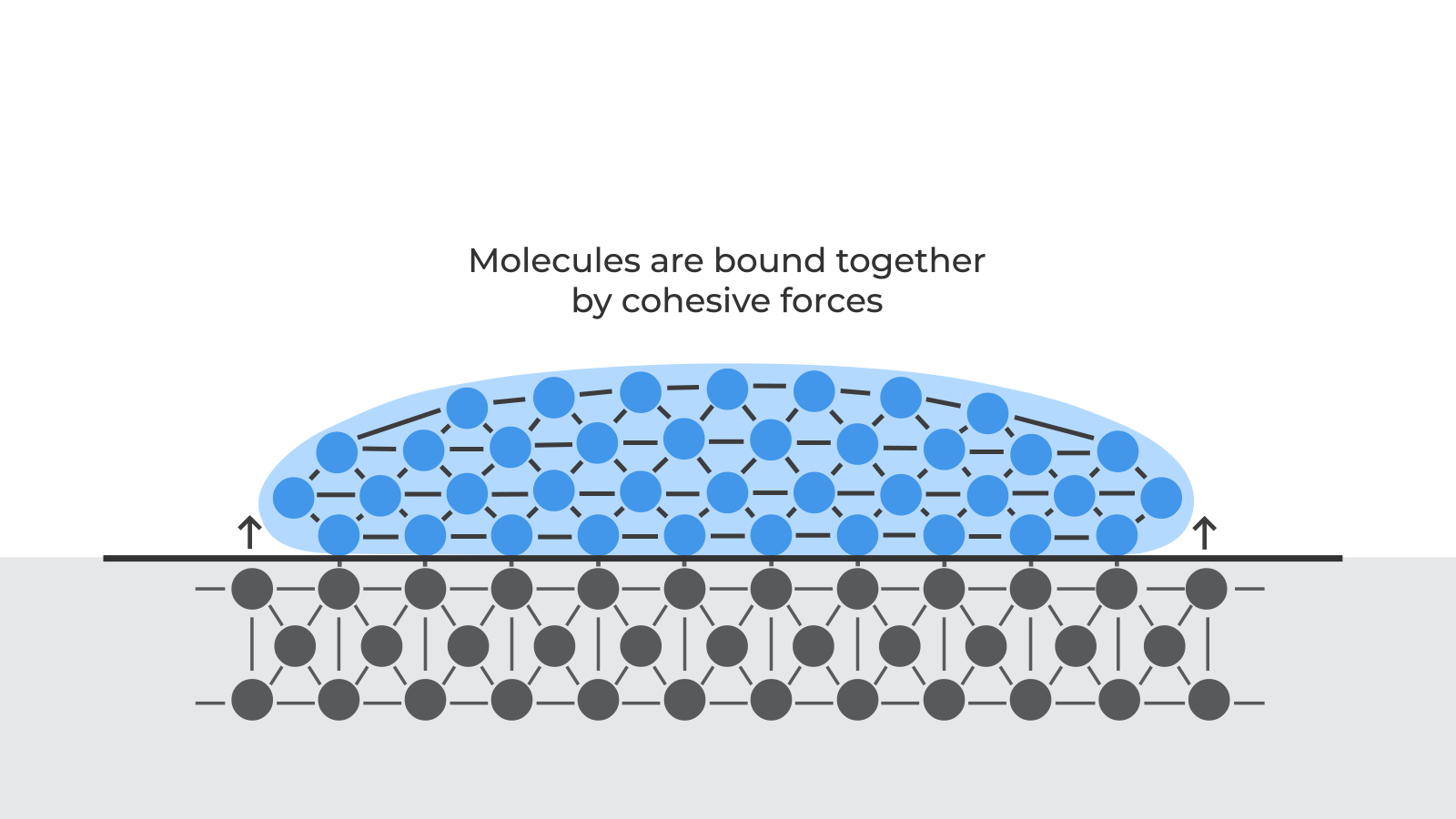
At a molecular level, materials bond together due to various cohesive forces. When parts are created, these cohesive forces are disrupted at the surface of the material because there are no molecules above the surface to interact with. This disruption leads to an excess of energy at the surface. This is what we call the surface free energy.
Surface free energy means that molecules are available to bond with other molecules. The higher the surface energy, the stronger the bond with other materials.
Surface preparation before adhesive bonding aims to increase the surface free energy of a material.
Removing surface contaminants is one of the best ways to improve adhesive bonding. Contaminants like dust and oils interfere with the adhesive bonding process because they bond with molecules on the surface of materials. They diminish the surface energy available for adhesive bonding and, as a result, the bond quality of adhesives.
Contaminant removal can be done with methods like laser cleaning, abrasive blasting, chemical cleaning, and manual degreasing.
Increasing surface roughness is not always necessary to obtain a good bond, but it can help obtain a stronger one. Surface roughness enhances bonding by increasing the contact area for adhesives to adhere to. The adhesive material fills the voids created by surface roughness, leading to improved bonding strength.
Different materials may or may not require roughening. Metals and plastics, for example, tend to have a lower surface free energy than other materials. For this reason, they can usually benefit most from surface roughening.
Surface roughening can be done with methods likes laser texturing, abrasive blasting, and chemical etching.
The chemical composition of a surface refers to the chemical elements that make up a surface as well as how the atoms are connected. It is possible to modify the chemical composition of a surface to create a more favorable surface for bonding.
Different treatments can introduce new elements on a surface, remove others, or break and rearrange the chemical bonds between atoms. Plasma treatments, laser texturing, and chemical processes can be used to modify the chemical composition of surfaces.
Laser surface treatments can remove surface contaminants, modify the surface roughness and structure, and introduce beneficial oxides to the surface. By promoting a strong bond from multiple angles, lasers provide a well-rounded surface preparation for adhesive bonding. They allow manufacturers to prepare surfaces in a single step.
During the laser process, the laser beam carries huge amounts of energy. Part of this energy is absorbed by the surface, causing it to heat up. This can be used to ablate contaminants and eject them from the surface. Since contaminants and metals are ablated at different temperatures, contaminants can be removed without damaging or texturing the underlying metal surface. If needed, laser systems can send more energy on the surface to clean and texture it at the same time.
Laser treatments have shown great results when preparing surfaces for structural adhesives like Loctite EA 9640 (and epoxy-based adhesive) and Betamate 4601 (a polyurethane-based adhesive), which are widely used for adhesive bonding applications in the automotive industry.
Beneficial Surface Oxides
During laser texturing, the plasma plume generated by the high temperature at the localized surface causes a chemical reaction with the ambient air. This creates a surface oxide layer that is beneficial for bonding in addition to the surface roughness that promotes adhesion. It is common belief that oxides are bad for adhesion, but research shows that this is not always the case. The ones created during laser texturing, for example, increase the surface free energy. While they can be removed by laser cleaning, studies show that removing them holds no benefits. | 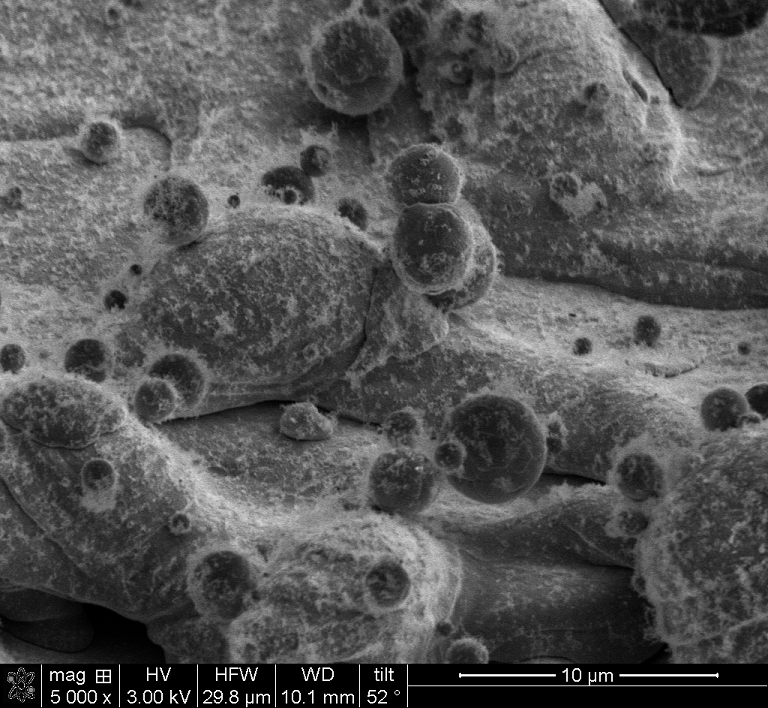 |
Mechanical abrasives are hard materials used to roughen or smooth surfaces through rubbing, grinding, or blasting. They can also remove most contaminants, including dust, dirt, and corrosion. While they help remove grease and oil, additional chemical degreasing steps are typically required to remove all contaminants.
It’s also important to note that mechanical abrasives leave dust behind. This dust originates from the abraded surface and from the abrasive media. This is why many manufacturers are moving away from mechanical abrasives like grit blasting, as dust can contaminate surfaces prior to bonding or find its way into parts.
Before abrading the surface, a degreasing step is typically required. Solvents like isopropanol or acetone are used to remove oil, dust, and grease residues. Then, sandblasting, wire brushing or other abrasives are used to remove surface deposits and increase the surface area (manual operators often use sandpaper). Finally, a degreasing step is required to remove loose particles.
Chemical processes can increase surface roughness by etching random patterns and remove a wide variety of surface contaminants. They can also change the chemical composition of the surface by introducing elements that promote bonding, or by breaking and rearranging the chemical bonds between atoms.
Chemical treatments, such as phosphoric acid anodizing and boric sulfuric acid anodizing, can form oxide films on surfaces that improve adhesion. These treatments are widely used in the aircraft and aerospace industry to prepare parts for bonding.
Through plasma, flame, or corona discharge, plasma treatments can clean, etch, and functionalize surfaces to increase the available surface energy and enable strong bonds.
Plasma treatments can remove a wide range of surface contaminants like oil, dust, electrolyte, and paint. However, metal oxides are usually more resistant to plasma and can leave carbonized residues that stick to the surface and are hard to remove.
During plasma treatments, new chemical groups are introduced in the surface. This is known as surface functionalization, and it creates new bonding sites for the adhesive.
For many manufacturers, the difference between plasma and laser is unclear. While plasma is an ionized gas, a laser beam is concentrated beam of light. Lasers are better at etching metals and lead to a higher shear strength. Lasers also have fewer mechanical limitations, allowing them to meet faster cycle times in high-volume production lines.
When we develop laser processes for our customers, we use a range of tests and measurements to validate the quality of the surface preparation. This ensures that we meet their specification requirements as well as choose the best laser parameters for optimal quality.
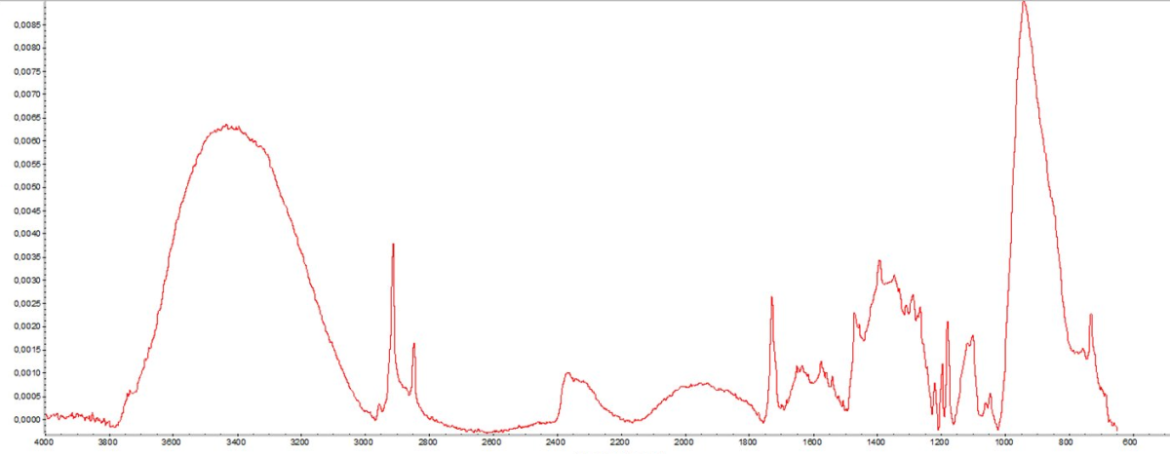
An FTIR measurement of a surface, showing the IR wavelength on the X-axis and its absorbance on the Z-axis. The absorbance of each wavelength provides information on the molecules present on the surface.
Spectrometers can be used to analyze the chemical composition of the surface. You can obtain precise information such as what contaminants are still present on the surface and how much, or what elements have been introduced or removed from the surface.
At Laserax, we use two types of spectrometers to analyze the chemical composition of the surface and understand the efficiency of our laser processes:
Wettability is a measure of how well a liquid will spread or bead up on the surface.
Wettability is a critical factor in adhesive bonding. The ability of a liquid to wet or not wet a surface can significantly influence the quality of the bond. Surface preparation is essential to control and tailor wettability.
Clean Surface – Low Contact Angle | Contaminated Surface – High Contact Angle |
 |  |
Computing the water contact angle (WCA) is one of the quickest and easiest ways to verify if you have a clean surface. By placing droplets from three different liquids on a surface (we use water, diiodomethane, and formadide), you can determine their contact angles and compute the surface free energy. A low contact angle indicates successful surface preparation, while a high contact angle indicates a contaminated surface.
Aging studies evaluate the long-term performance and durability of the adhesive bond under various environmental conditions. Aging tests are conducted to simulate the effects of time, temperature, humidity, and other environmental factors on the adhesive bond.
For example, the Jaguar JNS 30.03.35 cataplasm test evaluates the compatibility of the adhesive with the substrate by simulating extreme temperature changes. To perform the simulation, bonded joints are wrapped in cotton wool soaked in nanopure water. Placed in a sealed bag, the samples are then put into a heat chamber at 70°C (+158°F) for 7 days. On the 7th day, they are transferred into a cold chamber at -20°C (-4°F) for 16 hours. Finally, they are left to thaw and rest at room temperature, and a shear test is performed to evaluate the strength of the bond.
A wide range of aging tests can be performed to evaluate how the bond performs under different environmental conditions. The result of these tests can be critical in deciding between different surface preparation methods.
If you are considering laser surface preparation for adhesive bonding, you may want to read the following story:
We worked with a manufacturer who needed to prepare EV battery housings for adhesive bonding. They told us that they only wanted to clean the housing. They did not want to texture it even though it would increase the bond strength because, from previous experience, laser texturing was too long for their cycle time.
We convinced them otherwise.
Tests on their samples showed that our laser processing speed was much faster than what they were used to. This offered them the possibility to both clean and texture the surface and improve the bond strength without affecting cycle time.
As you probably deduced from this story, not all lasers are equal. The choice of laser components and parameters can be optimized to reach faster processing speeds. And this may be just what you need to make the difference.

Catherine holds a bachelor’s degree in Engineering Physics and a master's degree in Physics. She completed her master’s in partnership with Laserax to develop industrial solutions for the laser texturing of metallic surfaces. She is now the Applications Lab Supervisor at Laserax, where she oversees the team that tests and optimizes laser processes for clients.